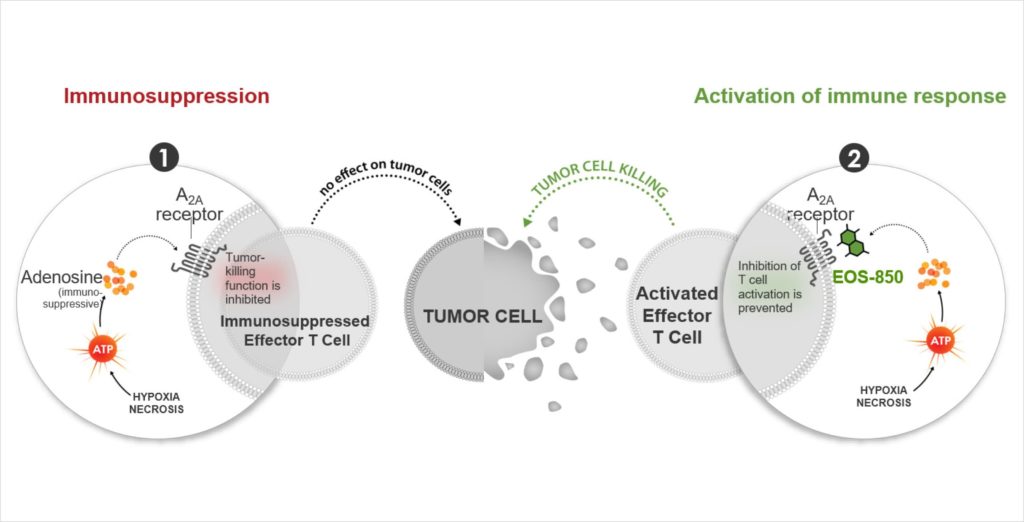
A treatment that targets immune cells’ primary adenosine receptor.
At iTeos we are pioneering the discovery and development of a new generation of highly differentiated immuno-oncology therapeutics.
EOS-448 is a human immunoglobulin antibody that we are developing to inhibit the immunosuppressive activity of TIGIT, a cell receptor expressed on multiple immune cells, including CD8+ T cells, natural killer (NK) cells, and T regulatory cells (Tregs).
Our differentiated development approach is driven by biologic rationale and clinical need utilizing our IO candidates in intra-portfolio combinations and with programs of external collaborators.
Latest press releases
iTeos Announces Clinically Meaningful Objective Response Rate Observed at Eve…
– Clinically meaningful objective response rate (ORR) of 63.3-76.7% observed with belrestotug + dostarlimab combinations, with confirmed ORR (cORR) at…
iTeos to Participate in Upcoming Investor Conferences
WATERTOWN, Mass. and GOSSELIES, Belgium, Aug. 28, 2024 (GLOBE NEWSWIRE) — iTeos Therapeutics, Inc. (Nasdaq: ITOS) (“iTeos”), a clinical-stage biopharmaceutical…